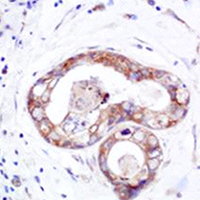
Expressions of ZNF436, β-catenin, EGFR, and CMTM5 in breast cancer and their clinical significances
Submitted: 13 August 2020
Accepted: 7 December 2020
Published: 20 January 2021
Accepted: 7 December 2020
Abstract Views: 1458
PDF: 595
Supplementary: 131
HTML: 11
Supplementary: 131
HTML: 11
Publisher's note
All claims expressed in this article are solely those of the authors and do not necessarily represent those of their affiliated organizations, or those of the publisher, the editors and the reviewers. Any product that may be evaluated in this article or claim that may be made by its manufacturer is not guaranteed or endorsed by the publisher.
All claims expressed in this article are solely those of the authors and do not necessarily represent those of their affiliated organizations, or those of the publisher, the editors and the reviewers. Any product that may be evaluated in this article or claim that may be made by its manufacturer is not guaranteed or endorsed by the publisher.
Similar Articles
- CarloAlberto Redi, Apoptosis - Methods and Protocols , European Journal of Histochemistry: Vol. 54 No. 1 (2010)
- G. Natale, E. Pompili, F. Biagioni, S. Paparelli, P. Lenzi, F. Fornai, Histochemical approaches to assess cell-to-cell transmission of misfolded proteins in neurodegenerative diseases , European Journal of Histochemistry: Vol. 57 No. 1 (2013)
- Yin Pan, Di Qiu, Shu Chen, Xiaoxue Han, Ruiman Li, High glucose inhibits neural differentiation by excessive autophagy via peroxisome proliferator-activated receptor gamma , European Journal of Histochemistry: Vol. 67 No. 2 (2023)
- Ermanna Turano, Federica Virla, Ilaria Scambi, Sylwia Dabrowska, Oluwamolakun Bankole, Raffaella Mariotti, Adipose mesenchymal stem cells-derived extracellular vesicles exert their preferential action in damaged central sites of SOD1 mice rather than peripherally , European Journal of Histochemistry: Vol. 68 No. 3 (2024)
- L Maccatrozzo, M Patruno, L Toniolo, C Reggiani, F Mascarello, Myosin Heavy Chain 2B isoform is expressed in specialized eye muscles but not in trunk and limb muscles of cattle , European Journal of Histochemistry: Vol. 48 No. 4 (2004)
- KH Cho, HS Lee, SK Ku, Changes in gastric endocrine cells in Balb/c mice bearing CT-26 carcinoma cells: an immunohistochemical study , European Journal of Histochemistry: Vol. 50 No. 4 (2006)
- P. Narducci, R. Bortul, R. Bareggi, V. Nicolin, Clathrin-dependent endocytosis of membrane-bound RANKL in differentiated osteoclasts , European Journal of Histochemistry: Vol. 54 No. 1 (2010)
- D. Stępiński, Immunofluorescent localization of ubiquitin and proteasomes in nucleolar vacuoles of soybean root meristematic cells , European Journal of Histochemistry: Vol. 56 No. 2 (2012)
- L.V. Renna, R. Cardani, A. Botta, G. Rossi, B. Fossati, E. Costa, G. Meola, Premature senescence in primary muscle cultures of myotonic dystrophy type 2 is not associated with p16 induction , European Journal of Histochemistry: Vol. 58 No. 4 (2014)
- Carla Loreto, Rosario Caltabiano, Adriana Carol Eleonora Graziano, Sergio Castorina, Claudia Lombardo, Vera Filetti, Ermanno Vitale, Giuseppe Rapisarda, Venera Cardile, Caterina Ledda, Venerando Rapisarda, Defense and protection mechanisms in lung exposed to asbestiform fiber: the role of macrophage migration inhibitory factor and heme oxygenase-1 , European Journal of Histochemistry: Vol. 64 No. 2 (2020)
<< < 42 43 44 45 46 47 48 49 50 51 > >>
You may also start an advanced similarity search for this article.

 https://doi.org/10.4081/ejh.2021.3173
https://doi.org/10.4081/ejh.2021.3173