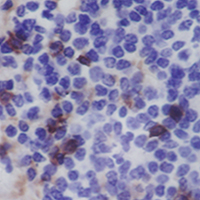
Characterization of PD-1/PD-L1 immune checkpoint expression in soft tissue sarcomas
Submitted: 21 November 2020
Accepted: 25 June 2021
Published: 2 July 2021
Accepted: 25 June 2021
Abstract Views: 2419
PDF: 700
HTML: 21
HTML: 21
Publisher's note
All claims expressed in this article are solely those of the authors and do not necessarily represent those of their affiliated organizations, or those of the publisher, the editors and the reviewers. Any product that may be evaluated in this article or claim that may be made by its manufacturer is not guaranteed or endorsed by the publisher.
All claims expressed in this article are solely those of the authors and do not necessarily represent those of their affiliated organizations, or those of the publisher, the editors and the reviewers. Any product that may be evaluated in this article or claim that may be made by its manufacturer is not guaranteed or endorsed by the publisher.
Similar Articles
- Michelle Steicke, Guang Yang, Tam Nguyen Dinh, Matthew Dunster-Jones, Owen Sargisson, Farah Ahmady, Jonathan Golledge, Yutang Wang, The penetration of methanol into bovine cardiac and hepatic tissues is faster than ethanol and formalin , European Journal of Histochemistry: Vol. 62 No. 1 (2018)
- Kazuhiko Hashimoto, Shunji Nishimura, Tomohiko Ito, Ryosuke Kakinoki, Masao Akagi, Immunohistochemical expression and clinicopathological assessment of PD-1, PD-L1, NY-ESO-1, and MAGE-A4 expression in highly aggressive soft tissue sarcomas , European Journal of Histochemistry: Vol. 66 No. 2 (2022)
- A. Porzionato, M. M. Sfriso, V. Macchi, A. Rambaldo, G. Lago, L. Lancerotto, V. Vindigni, R. De Caro, Decellularized omentum as novel biologic scaffold for reconstructive surgery and regenerative medicine , European Journal of Histochemistry: Vol. 57 No. 1 (2013)
- M.L. Escobar Sánchez, O.M. EcheverrÃa MartÃnez, G.H. Vázquez-Nin, Immunohistochemical and ultrastructural visualization of different routes of oocyte elimination in adult rats , European Journal of Histochemistry: Vol. 56 No. 2 (2012)
- M. Lehmann, F. Martin, K. Mannigel, K. Kaltschmidt, U. Sack, U. Anderer, Three-dimensional scaffold-free fusion culture: the way to enhance chondrogenesis of in vitro propagated human articular chondrocytes , European Journal of Histochemistry: Vol. 57 No. 4 (2013)
- Martin Boháč, Ľuboš Danišovič, Ján Koller, Jana Dragúňová, Ivan Varga, What happens to an acellular dermal matrix after implantation in the human body? A histological and electron microscopic study , European Journal of Histochemistry: Vol. 62 No. 1 (2018)
- Kazuhiko Hashimoto, Shunji Nishimura, Yu Shinyashiki, Tomohiko Ito, Ryosuke Kakinoki, Masao Akagi, Clinicopathological assessment of PD-1/PD-L1 immune checkpoint expression in desmoid tumors , European Journal of Histochemistry: Vol. 67 No. 2 (2023)
- J. Rieger, S. Twardziok, H. Huenigen, R. M. Hirschberg, J. Plendl, Porcine intestinal mast cells. Evaluation of different fixatives for histochemical staining techniques considering tissue shrinkage , European Journal of Histochemistry: Vol. 57 No. 3 (2013)
- A.M. Schläfli, S. Berezowska, O. Adams, R. Langer, M.P. Tschan, Reliable LC3 and p62 autophagy marker detection in formalin fixed paraffin embedded human tissue by immunohistochemistry , European Journal of Histochemistry: Vol. 59 No. 2 (2015)
- A.C. Croce, G. Bottiroli, Autofluorescence spectroscopy and imaging: a tool for biomedical research and diagnosis , European Journal of Histochemistry: Vol. 58 No. 4 (2014)
You may also start an advanced similarity search for this article.
Publication Facts
Metric
This article
Other articles
Peer reviewers
5
2.4
Reviewer profiles N/A
Author statements
Author statements
This article
Other articles
Data availability
N/A
16%
External funding
N/A
32%
Competing interests
N/A
11%
Metric
This journal
Other journals
Articles accepted
57%
33%
Days to publication
222
145
- Academic society
- N/A
- Publisher
- PAGEPress Publications, Pavia, Italy

 https://doi.org/10.4081/ejh.2021.3203
https://doi.org/10.4081/ejh.2021.3203