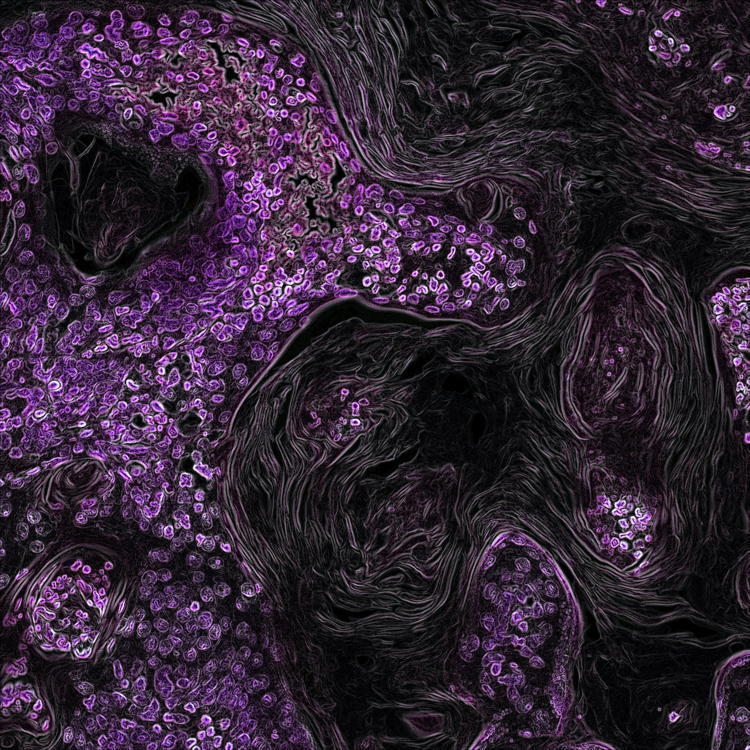
Seasonal expressions of GPR41 and GPR43 in the colon of the wild ground squirrels (Spermophilus dauricus)
Submitted: 23 October 2021
Accepted: 2 January 2022
Published: 21 January 2022
Accepted: 2 January 2022
Abstract Views: 1150
PDF: 558
HTML: 27
HTML: 27
Publisher's note
All claims expressed in this article are solely those of the authors and do not necessarily represent those of their affiliated organizations, or those of the publisher, the editors and the reviewers. Any product that may be evaluated in this article or claim that may be made by its manufacturer is not guaranteed or endorsed by the publisher.
All claims expressed in this article are solely those of the authors and do not necessarily represent those of their affiliated organizations, or those of the publisher, the editors and the reviewers. Any product that may be evaluated in this article or claim that may be made by its manufacturer is not guaranteed or endorsed by the publisher.
Similar Articles
- CarloAlberto Redi, Next-generation nutritional biomarkers to guide better health care , European Journal of Histochemistry: Vol. 60 No. 2 (2016)
- M. Scimeca, A. Orlandi, I. Terrenato, S. Bischetti, E. Bonanno, Assessment of metal contaminants in non-small cell lung cancer by EDX microanalysis , European Journal of Histochemistry: Vol. 58 No. 3 (2014)
- Suwei Dong, Yanbin Xiao, Ziqiang Zhu, Xiang Ma, Zhuohui Peng, Jianping Kang, Jianqiang Wang, Yunqing Wang, Zhen Li, Metformin sensitises osteosarcoma to chemotherapy via the IGF-1R/miR-610/FEN1 pathway , European Journal of Histochemistry: Vol. 67 No. 2 (2023)
- L. Ragionieri, M. Botti, F. Gazza, C. Sorteni, R. Chiocchetti, P. Clavenzani, L. Bo, R. Panu, Localization of peripheral autonomic neurons innervating the boar urinary bladder trigone and neurochemical features of the sympathetic component , European Journal of Histochemistry: Vol. 57 No. 2 (2013)
- J. Xue, H. Chen, L. Diao, X. Chen, D. Xia, Expression of Caveolin-1 in tongue squamous cell carcinoma by quantum dots , European Journal of Histochemistry: Vol. 54 No. 2 (2010)
- Lu Liu, Zhankui Liu, Liu Yang, Xue Wu, Jiaying Zhu, Lili Liu, Yang Liu, Lobetyolin suppressed lung cancer in a mouse model by inhibiting epithelial-mesenchymal transition , European Journal of Histochemistry: Vol. 66 No. 3 (2022)
- Xiaoyan Chu, Jun Lou, Yun Yi, Linlin Zhong, Ouping Huang, Knockdown of ARHGAP30 inhibits ovarian cancer cell proliferation, migration, and invasiveness by suppressing the PI3K/AKT/mTOR signaling pathway , European Journal of Histochemistry: Vol. 67 No. 2 (2023)
- M Malatesta, B Baldelli, S Battistelli, C Tiberi, E Manuali, M Biggiogera, Reversibility of hepatocyte nuclear modifications in mice fed on genetically modified soybean , European Journal of Histochemistry: Vol. 49 No. 3 (2005)
- A.T.R. Clark, V.M.L. Guimaraes da Costa, L. Bandeira Costa, C.L. Bezerra Cavalcanti, M.J.B. de Melo Rego, E.I.C. Beltrao, Differential expression patterns of N-acetylglucosaminyl transferases and polylactosamines in uterine lesions , European Journal of Histochemistry: Vol. 58 No. 2 (2014)
- F. Frontalini, D. Curzi, F.M. Giordano, J.M. Bernhard, E. Falcieri, R. Coccioni, Effects of lead pollution on Ammonia parkinsoniana (foraminifera): ultrastructural and microanalytical approaches , European Journal of Histochemistry: Vol. 59 No. 1 (2015)
<< < 5 6 7 8 9 10 11 12 13 14 > >>
You may also start an advanced similarity search for this article.

 https://doi.org/10.4081/ejh.2022.3351
https://doi.org/10.4081/ejh.2022.3351