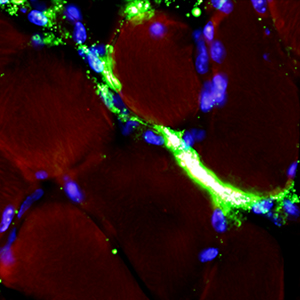
An ex vivo experimental system to track fluorescent nanoparticles inside skeletal muscle
Submitted: 7 November 2022
Accepted: 14 December 2022
Published: 22 December 2022
Accepted: 14 December 2022
Abstract Views: 789
PDF: 469
HTML: 28
HTML: 28
Publisher's note
All claims expressed in this article are solely those of the authors and do not necessarily represent those of their affiliated organizations, or those of the publisher, the editors and the reviewers. Any product that may be evaluated in this article or claim that may be made by its manufacturer is not guaranteed or endorsed by the publisher.
All claims expressed in this article are solely those of the authors and do not necessarily represent those of their affiliated organizations, or those of the publisher, the editors and the reviewers. Any product that may be evaluated in this article or claim that may be made by its manufacturer is not guaranteed or endorsed by the publisher.
Similar Articles
- E Brunelli, I Perrotta, A Bonacci, S Tripepi, Differential expression of aquaporin 3 in Triturus italicus from larval to adult epidermal conversion , European Journal of Histochemistry: Vol. 51 No. 1 (2007)
- CarloAlberto Redi, G-quadruplex DNA - Methods and protocols , European Journal of Histochemistry: Vol. 54 No. 3 (2010)
- Carlo Alberto Redi, Adipose-derived stem cells - Methods and protocols , European Journal of Histochemistry: Vol. 55 No. 3 (2011)
- D. Ami, M. Di Segni, M. Forcella, V. Meraviglia, M. Baccarin, S.M. Doglia, G. Terzoli, Role of water in chromosome spreading and swelling induced by acetic acid treatment: a FTIR spectroscopy study , European Journal of Histochemistry: Vol. 58 No. 1 (2014)
- F. Cau, E. Pisu, C. Gerosa, G. Senes, F. Ronchi, C. Botta, E. Di Felice, F. Uda, V. Marinelli, G. Faa, V. Fanos, C. Moretti, D. Fanni, Interindividual variability in the expression of surfactant protein A and B in the human lung during development , European Journal of Histochemistry: Vol. 60 No. 3 (2016)
- Filippo Vernia, Tiziana Tatti, Stefano Necozione, Annalisa Capannolo, Nicola Cesaro, Marco Magistroni, Marco Valvano, Simona Pompili, Roberta Sferra, Antonella Vetuschi, Giovanni Latella, Is mastocytic colitis a specific clinical-pathological entity? , European Journal of Histochemistry: Vol. 66 No. 4 (2022)
- M Biggiogera, L Cocco, FA Manzoli, Structure and function of the cell nucleus , European Journal of Histochemistry: Vol. 48 No. 1 (2004)
You may also start an advanced similarity search for this article.

 https://doi.org/10.4081/ejh.2023.3596
https://doi.org/10.4081/ejh.2023.3596